My research investigates how organisms navigate fluctuating and often contrasting demands on life-history traits resulting from climate change under different ecological contexts (e.g., predation risk).
I approach ecological questions through a mixture of long-term field-based research, lab experiments, and mathematical modeling to refine and develop new theories of how organisms are and will continue to respond to climate change effects (e.g., drought, thermal regimes).
I approach ecological questions through a mixture of long-term field-based research, lab experiments, and mathematical modeling to refine and develop new theories of how organisms are and will continue to respond to climate change effects (e.g., drought, thermal regimes).
|
Current CV
|
| ||||||
Thermal Ecology
All life happens at some temperature, but happens differently at different temperatures.
|
All life is comprised of tiny particles that interact with each other (e.g., enzymes, substrates). The speed and likelihood of their interactions are directly affected by changes in temperature. Indeed, scientists have long recognized the impact of temperature on experiments and you'll scarcely find a study where temperature was not controlled or recorded. Within an organism, changes in temperature affect metabolism (how quickly organisms process materials), development rate (e.g., how quickly an egg changes into an embryo and an adult), and can even lead to changes in development that determine sex (e.g., turtles, crocodilians). For ectotherms, whose body temperature largely reflects that of their environment, changes to their thermal environment affect nearly all aspects of their ecology.
Ectotherms are by far the most abundant life on Earth and play critical roles in the functioning of every ecosystem. For example, insects are estimated to make up the majority of animal mass on land and number somewhere around 10 quintillion individuals (that's 1 followed by nineteen 0's) (Smithsonian BugInfo). In the oceans, ectotherms such as phytoplankton transfer an estimated 10 gigatons of carbon from the atmosphere to the deep ocean annually (NASA Earth Observatory). Choose any living thing on Earth... it either is an ectotherm or depends on ectotherms for its survival. While the general effects of temperature on organisms are fairly well understood, temperature is not the only factor that affects organismal physiology. Thermal ecology places the temperature dependence of biological processes within the context of how organisms interact with each other and their environment. My work in thermal ecology generally focuses on how predation and the threat of predation (predation risk) affect the temperature dependence of population growth and life-history strategies (such as how many offspring to have and how soon to have them, etc.) |
Much like molecules in a cell, the speeds of these Cyclops (big and orange) and Paramecium (smaller and thin) change with temperature. Because speed changes encounter rates between predators and prey, temperature effects on speed likewise affect predator-prey dynamics.
The sex of this hatching diamondback terrapin was determined by the range of temperatures it experienced during a critical phase of its development inside its egg.
|
Obligatory pictures of example ectotherms largely biased towards vertebrates (clockwise from top left: bumble bee in a water lily flower - Aiken, SC; green tree frog in a summer chorus - Aiken, SC; green anole, cold and unhappy at being woken up - Lincolnton, GA; juvenile rainbow snake from a Carolina Bay in Aiken, SC; spring salamander from Great Smokey Mountains National Park, TN).
Thermal Performance Curves (TPC)One way to quantify the capacity of organisms to weather changing thermal regimes is with thermal performance curves (TPCs), which depict the change in a performance metric (e.g., sprint speed, metabolism, population growth rate) across some range of temperatures.
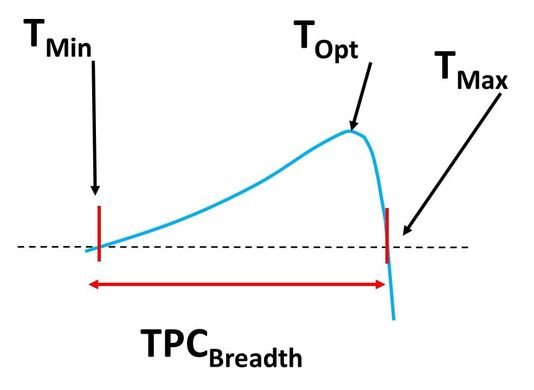
Thermal performance curves for population growth rate and metabolism generally show an increase in performance across a warming gradient prior to reaching an optimal temperature and then declining (often at a precipitous rate). Certain points along the TPC are of special interest to ecologists because they denote important transitions such as: the lower temperature where the metric approaches 0 (Tmin), the upper temperature at which the metric approaches 0 (Tmax), and the temperature at which the metric reaches its maximum (Topt). Other descriptors of interest quantify the breadth over which the metric is positive (TPC Breadth), or the position of an organism on the curve at a given moment (e.g., thermal safety margin). |
Example thermal performance curve. The axis labels are omitted for visual clarity, but temperature would be on the x-axis and the blue line would show the nonlinear change in a performance metric such as population growth rate, sprint speed, or metabolism. The four metrics (Tmin, Topt, Tmax, and TPC Breadth) are described in the text.
|
Predation Effects on TPCs
Predation changes r TPCs
|
While TPCs provide a tool for tying the fitness of ectotherms to their thermal environment, most predictive approaches assume TPCs are invariant within a species or group. The problem with this assumption is that many temperature dependent traits are also highly sensitive to ecological context such as predation.
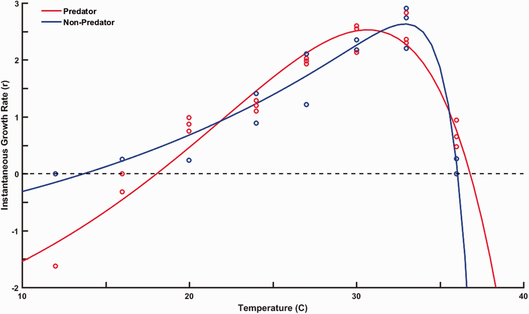
Indeed, our work shows that copepod predation readily changes the shape of r TPCs for Paramecium aurelia (Luhring and DeLong 2016) by:
These findings illustrate not only that predation interacts with temperature to change fitness, but that TPCs are plastic and change under varying ecological contexts. |
Thermal Performance Curves for population growth rate of the protist Paramecium aurelia change with predation.
(Figure 1; Luhring and DeLong 2016) |
Phenotypically plastic responses are temperature dependent (Dragons and fleas - Part I)
Odonate larvae (juvenile dragonflies and damselflies) are major aquatic predators of both invertebrates and vertebrates in freshwater ecosystems. Several potential prey of odonate larvae such as water fleas (one being eaten in this video) and amphibians show phenotypically plastic responses to the presence of olfactory cues from these predators. Video courtesy of Stella Uiterwaal, UNL.
While we knew that r TPCs changed with respect to predation (Luhring and DeLong 2016), it was unclear how underlying traits were changing with temperature and predation. However, given the short time-frame of our experiment we were inclined to believe that these responses were due to phenotypic plasticity and not evolution. Indeed, many organisms alter their life-histories in response to predation through phenotypic plasticity (non-genetic changes in traits such as changing the size at maturation).
To investigate how phenotypically plastic changes in life-history traits could change r TPCs, I collaborated with the Cressler lab to work with a clonal population of Daphnia magna (a species of water flea - a type of freshwater crustacean). In this experiment, we presented predation cues to individual Daphnia magna clones at varying times during their development (four treatments: no cue, early exposure, late exposure, constant exposure) across a 22°C range of temperature from 11-33°C.
Not only did r TPCs change with exposure to predation risk, but the timing of predation exposure changed the manner in which they changed (Luhring et al., 2019 - Oecologia). Differences in r TPC shapes among the four treatments were were caused by uneven responses of life-history traits across temperature that were further influenced cue timing (e.g., D. magna accelerate reproductive schedules under predation risk, but are less able to do so at colder temperatures if exposed later in ontogeny). In other words, phenotypically plastic responses to predation risk were temperature-dependent. (Luhring et al., 2019 - Oecologia) Link to Article (contact me for a PDF via email or research gate)
To investigate how phenotypically plastic changes in life-history traits could change r TPCs, I collaborated with the Cressler lab to work with a clonal population of Daphnia magna (a species of water flea - a type of freshwater crustacean). In this experiment, we presented predation cues to individual Daphnia magna clones at varying times during their development (four treatments: no cue, early exposure, late exposure, constant exposure) across a 22°C range of temperature from 11-33°C.
Not only did r TPCs change with exposure to predation risk, but the timing of predation exposure changed the manner in which they changed (Luhring et al., 2019 - Oecologia). Differences in r TPC shapes among the four treatments were were caused by uneven responses of life-history traits across temperature that were further influenced cue timing (e.g., D. magna accelerate reproductive schedules under predation risk, but are less able to do so at colder temperatures if exposed later in ontogeny). In other words, phenotypically plastic responses to predation risk were temperature-dependent. (Luhring et al., 2019 - Oecologia) Link to Article (contact me for a PDF via email or research gate)
Trade-offs are temperature dependent (Dragons and fleas - Part II)
|
Daphnia magna produce clutches of live offspring that they release from a special brood pouch. In addition to resource availability, Daphnia mothers are limited by how much offspring volume they can "pack" in their brood pouch (known as a packing constraint). Thus, two factors affect the total resource available to parse among their offspring - resource availability and parental size.
|
|
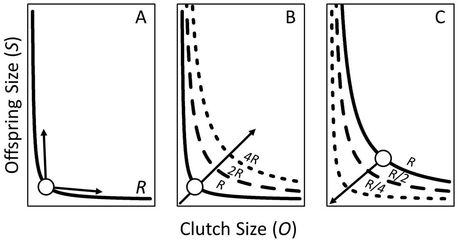
Some organisms respond to predation risk by altering their growth or reproductive strategies (e.g., timing of reproduction or the size and number of offspring). However, some of these adjustments are constrained whereby an increase in one trait requires a decrease in another (e.g., a trade-off). Offspring size and number are one such well-known example of a trade-off in life-history. Given a fixed energy budget, increases in offspring number can come about only through a decrease in their average size (and vice versa) - frequently referred to as the "size-number trade-off." Species under size-independent predation risk (e.g., all sizes are equally vulnerable to predation) or in systems where mortality risk is higher for larger individuals, producing a higher number of smaller offspring is generally a better strategy. This is because increasing offspring size is less advantageous than creating more offspring (there is a high per individual mortality risk.... so make more babies!).
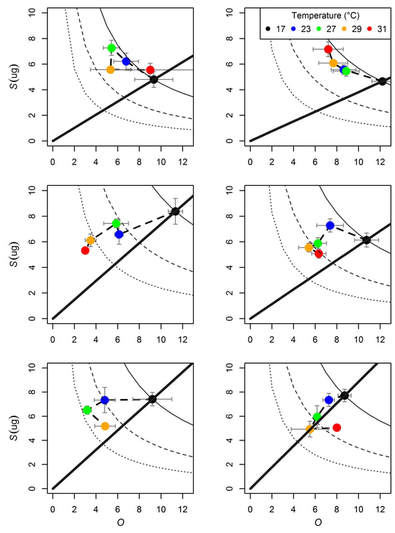
As you probably guessed, in addition to showing phenotypically plastic responses to predation risk - the amount of energy available to parse among offspring, body size, and clutch size are all temperature dependent. Using data from the above experiment, we showed that predation risk affected adult size at reproduction and temperature affected both timing of and size at reproduction (all as expected). After accounting for the timing of and size at reproduction, we likewise found that temperature interacts with predation risk to alter offspring size and number across the first three clutches (we used the first three clutches because most treatments and temperatures had at least three clutches across the experiment). However, interesting patterns emerged as we started looking at how temperature moved clutches around in size-number space (and how that pattern changed across clutches). In the first clutch, warmer temperatures moved clutches along the trade-off curve towards fewer larger offspring with the pattern being less noisy in the presence of predation cues (top row). As the experiment progressed and clutches were produced after longer exposure to TPC temperatures, clutches were consistently reduced in total R with increasing temperature (moving across the dashed lines). However, predation risk resulted in a lesser relative decrease in R and offspring number with increased temperatures (in most cases only approaching the 1/2 isocline) and by the third clutch showed a higher propensity than the control to sacrifice offspring size to maintain higher clutch sizes. |
Given a fixed resource (R), the number of offspring (O) cannot change without a change in their size (S) (panel A). However, if the amount of R available to parse among offspring increases (panel B) or decreases (panel C) O and S can simultaneously increase or decrease as their trade-off isoclines move in space (dashed lines show the new possible combinations of O and S given increases or decreases in R). (Figure 1 in Luhring et al., 2018 Ecology and Evolution)
Daphnia magna without exposure to predation cues (control - left column) and those exposed to predation cues throughout the experiment (constant exposure - right column) differed in how strongly temperature shifted their allocation strategies of size vs. number of offspring across their first three clutches (from top to bottom). Dashed lines indicate 1/2 and 1/4 total R of that in the 17°C group. The solid line runs from the 17°C group through the origin to indicate a 1:1 change in offspring size and number. (Figure 3 Luhring et al., 2018 Ecology and Evolution).
|
Body Size Ecology and Evolution
Body size affects everything from what you eat, what eats you, your metabolism, and whether or not you fit in a trashcan.
Environmental Body Size Selection
|
Drought tolerance of aquatic species in isolated wetlands is strongly related to body size. In many cases, it appears that larger individuals generally do worse during droughts because they have higher overall metabolic demands and starve to death faster than smaller individuals. Prior research (Snodgrass et al. 1999) showed that two species of closely related aquatic salamanders while being found in the same landscape, were generally not found in the same wetlands. They hypothesized that hydroperiod (the length of time a wetland retains water during the year) and competition were the primary drivers of their uncommon cohabitation.
Since 2006, I have tracked an individually-marked population of the larger species (Siren lacertina) (and another large aquatic salamander - Amphiuma means) at an isolated wetland in Aiken, SC (in collaboration with the Savannah River Ecology Lab). During this time, repeated droughts hit the SE US and the wetland completely dried for months at a time. Although they are aquatic and they aren't great at moving overland (shown by my former REU student Dr. Chris Schalk: [PDF]) sirenids persist during droughts by burying in the mud and aestivating just like lungfish! Using known size-specific growth rates (Luhring 2008), I showed that gaps in size-distributions evident for several years following severe droughts in 1989 (data from Tony Tucker), 2001, and 2011 were caused by drought-induced mortality of the smallest size classes (Luhring and Holdo 2015). We attribute this reversal in drought size selection (relative to other species where larger individuals are the ones that blink out) to the fact that greater sirens don't just get bigger, they get bulkier. This added bulk (lipids) provides added fuel to survive droughts and larger individuals have more of it. Considering they can lose over 80% of their body mass and still survive (that's like a 200lb human losing 160lbs), sirens are master fasters and superbly suited to life in variable habitats. Several other ongoing and completed projects associated with this study (both by myself and undergraduate collaborators) will be described more in a future page. An individually-marked (PIT tag) Siren lacertina from my long-term field site (Dry Bay, Aiken, SC). There have been more than 800 captures of individuals (294 recaptures) that altogether measure longer than a quarter mile and nearly 700lbs. The current record for largest individual is 1.5 kg (3.3 lbs) and 80.3cm (2.6 ft).
|
Period droughts are a regular occurance in isolated wetlands.
Aquatic organisms in drying habitats can either die (such as these toad tadpoles in a drying wetland), emigrate to other wetlands or stay in place and wait out the drought.
Three different sizes, all one species. Greater sirens start off pretty small (the one on the right is ~1-2 years old) but get big and delay putting a substantial investment into reproduction until they get large enough to survive droughts.
|
Eco-Evolutionary Dynamics of Body Size Evolution
While many of us have ecology and evolution filled away in discrete file drawers of our brains, they are intricately interwoven and inseparable in nature. One glaring example is seen in the interactions of organisms in functioning food webs and the many effects that body size has on these interactions.
Changes to body size affect eco-evolutionary dynamics of food webs because selection acts strongly on body size, body size strongly affects species interactions, and ecological and evolutionary processes occur on similar timescales. Predation (including human harvest of commercial and game species) often drives evolution of body size toward smaller sizes by selecting for earlier maturation because having offspring earlier reduces the likelihood of being predated before reproduction.
We use Gillespie eco-evolutionary models (DeLong and Gibert 2016; DeLong and Luhring 2018) for investigating eco-evolutionary dynamics of body size and life-history trait evolution because they enable trait distributions to change as individuals are born or die and instantaneously update trait-dependent event probabilities (e.g., predation event, birth).
Recent efforts investigatied how eco-evolutionary dynamics of body size evolution transfer across trophic levels under varying scenarios of predator removal and introduction (Luhring and DeLong 2020).
Changes to body size affect eco-evolutionary dynamics of food webs because selection acts strongly on body size, body size strongly affects species interactions, and ecological and evolutionary processes occur on similar timescales. Predation (including human harvest of commercial and game species) often drives evolution of body size toward smaller sizes by selecting for earlier maturation because having offspring earlier reduces the likelihood of being predated before reproduction.
We use Gillespie eco-evolutionary models (DeLong and Gibert 2016; DeLong and Luhring 2018) for investigating eco-evolutionary dynamics of body size and life-history trait evolution because they enable trait distributions to change as individuals are born or die and instantaneously update trait-dependent event probabilities (e.g., predation event, birth).
Recent efforts investigatied how eco-evolutionary dynamics of body size evolution transfer across trophic levels under varying scenarios of predator removal and introduction (Luhring and DeLong 2020).
Publications
(All listed publications available on request; Undergraduate Collaborators Underlined)
- Ward, KJ, EA Stybr, SEM Burkhead, TM Luhring. (2023) No free refills: previously dried wetlands function differently than undried wetlands following refill, with implications for vertebrate fitness. Journal of Freshwater Biology 68:1614-1626
- Luhring, TM, LS Wszola, GM Connette, CM Schalk (2022) Droughts reduce growth and increase demographic vulnerability to drying events for an ectotherm with size-dependent estivation ability. Journal of Herpetology 56:521-527. DOI: 10.1670/21-084
*Highlighted in BioOne Complete’s Top & Trending Research April 2023: https://bioone.org/top-and-trending-april-2023 - Burkhead, SEM, CS Streid, JT Wright, SA Bristow, PL Hoang, JL Oettle, A Pham, S Pulliam, DR Mardis, EA Stybr, KJ Ward, TM Luhring (2022) Aquatic systems of the Wichita State University Biological Field Station: Youngmeyer Ranch, Elk County, Kansas. Transactions of the Kansas Academy of Science 125:109-118
- Ward, KJ, CS Streid, JT Wright, TM Luhring. (2022) Regina grahamii (Graham’s Crayfish Snake). Herpetological Review 53:264
- Little, CJ, M Rizzuto, TM Luhring, JD Monk, RJ Nowicki, RE Paseka, J Stegen, CC Symons, FB Taub, JD Yen (2022) Movement with meaning: integrating information into meta-ecology. Oikos 2022:e08892
- Sabal, MC, MS Boyce, CL Charpentier, NB Furey, TM Luhring, HW Martin, MC Melnychuk, RB Srygley, CM Wagner, AJ Wirsing, RC Ydenberg, EP Palkovacs (2021) Predation landscapes influence migratory prey ecology and evolution. Trends in Ecology and Evolution 36:737-749
- Luhring, TM, JP DeLong (2020) Trophic cascades alter eco-evolutionary dynamics and body size evolution. Proceedings of the Royal Society B 287:20200526. DOI: 10.1098/rspb.2020.0526
- Hume, JB, TM Luhring, CM Wagner (2020). Push, pull, or push-pull? An alarm cue better guides sea lamprey towards capture devices than a migrating pheromone during the reproductive migration. Biological Invasions 22:2129-2142 DOI: 10.1007/s10530-020-02242-4
- Uiterwaal, S, I Lagerstrom, TM Luhring, ME Salsbery, JP DeLong (2020). Trade-offs between morphology and thermal niches mediate adaptation in response to competing selective pressures. Ecology and Evolution DOI: 10.1002/ece3.5990
- Luhring, TM, JM Vavra, CE Cressler, JP DeLong (2019). Phenotypically plastic responses to predation risk are temperature dependent. Oecologia 191:709-719. DOI: 10.1007/s00442-019-04523-9
- DeLong, JP, G Bachman, JP Gibert, TM Luhring, K Montooth, K Neyer, B Reed (2018). Habitat, latitude, and body mass influence the temperature dependence of metabolic rate. Biology Letters. 14:20180442. DOI: 10.1098/rsbl.2018.0442
- Luhring, TM, JM Vavra, CE Cressler, JP DeLong (2018). Predators modify the thermal dependence of life-history trade-offs. Ecology and Evolution. DOI: 10.1002/ece3.4381
- Relyea, RA, et al. (15th of 25 authors) (2018). Phylogenetic patterns of trait plasticity evolution: Insights from amphibian embryos. Evolution. DOI: 10.1111/evo.13428
- DeLong, JP, TM Luhring (2018). Size-dependent predation and correlated life history traits alter eco-evolutionary dynamics and selection for faster individual growth. Population Ecology DOI: 10.1007/s10144-018-0608-7
- Luhring, TM, JP DeLong (2017). Scaling from metabolism to population growth rate to understand how acclimation temperature alters thermal performance. Integrative and Comparative Biology https://doi.org/10.1093/icb/icx041
- Luhring, TM, JP DeLong, RD Semlitsch (2017). Stoichiometry and life- history interact to determine the magnitude of cross-ecosystem element and biomass fluxes. Frontiers in Microbiology 8:814 DOI: 10.3389/fmicb.2017.00814 (Special Research Topic: Progress in Ecological Stoichiometry)
- DeLong, JP, JP Gibert, TM Luhring, G Bachman, B Reed, A Neyer, KL Montooth (2017). The combined effects of reactant kinetics and enzyme stability explain the temperature dependence of metabolic rates. Ecology and Evolution DOI: 10.1002/ece3.2955
- Luhring, TM, TD Meckley, JB Hume, N Johnson, M Siefkes, CM Wagner (2016). A semelparous fish continues upstream migration when exposed to alarm cue, but adjusts movement speed and timing. Animal Behaviour 121:41-51 (DOI: 10.1016/j.anbehav.2016.08.007)
- Wagner, CM, KE Kierczynski, JB Hume, TM Luhring (2016). Exposure to a putative alarm cue reduces downstream drift in larval sea lamprey (Petromyzon marinus) in the laboratory. Journal of Fish Biology 89:1897-1904 (DOI: 10.1111/jfb.13095)
- Luhring, TM, JP DeLong (2016). Predation changes the shape of thermal performance curves for population growth rate. Current Zoology 2016:1-5 (PDF) (DOI: 10.1093/cz/zow045)
- Luhring, TM, GC Connette, CM Schalk (2016). Trap characteristics and species morphology explain size-biased sampling of two salamander species. Amphibia-Reptilia 37:79-89 (DOI: 10.1163/15685381-00003034)(SREL Reprint #3343)(PDF)
- Hume, JB, TD Meckley, NS Johnson, TM Luhring, MJ Siefkes, CM Wagner (2015). Application of putative alarm cue hastens the arrival of invasive sea lamprey (Petromyzon marinus) at a trapping location. Canadian Journal of Fisheries and Aquatic Sciences 72:1799-1806. (DOI: 10.1139/cjfas-2014-0535) (PDF)
- Luhring, TM, RM Holdo (2015). Trade-offs between age at reproduction and body size; drought survival in stochastic aquatic environments. Oecologia 178:723-732. (DOI: 10.1007/s00442-015-3270-1) (PDF) (Supplementary Material) (SREL Reprint #3323)
- Ousterhout, B*, TM Luhring*, RD Semlitsch (2014). No evidence of natal habitat preference induction in juveniles with complex life-histories. Animal Behaviour 93:237-242 (*Equal Authorship) (PDF)
- Luhring, TM, JW Gibbons, PW Gibbons (2013). Siren sp. predation. Herpetological Review 44:491-492. (PDF) (SREL Reprint #3256)
- Luhring, TM (2013). Auditory surveys. In: GJ Graeter, KA Buhlmann, LR Wilkinson, JW Gibbons, eds. Inventory and Monitoring: Recommended Techniques for Reptiles and Amphibians, with application to the United States and Canada. PARC Technical Report. Aiken, South Carolina.
- Luhring, TM (2013). Leaf-litterbag surveys. In: GJ Graeter, KA Buhlmann, LR Wilkinson, JW Gibbons, eds. Inventory and Monitoring: Recommended Techniques for Reptiles and Amphibians, with application to the United States and Canada. PARC Technical Report. Aiken, South Carolina.
- Luhring, TM (2013). Trashcan traps. In: GJ Graeter, KA Buhlmann, LR Wilkinson, JW Gibbons, eds. Inventory and Monitoring: Recommended Techniques for Reptiles and Amphibians, with application to the United States and Canada. PARC Technical Report. Aiken, South Carolina.
- Reed, RN, TM Luhring, AD Tucker (2013). Appendix III: Determination of age, sex and reproductive condition. In: GJ Graeter, KA Buhlmann, LR Wilkinson, JW Gibbons, eds. Inventory and Monitoring: Recommended Techniques for Reptiles and Amphibians, with application to the United States and Canada. PARC Technical Report. Aiken, South Carolina.
- Luhring, TM, Z.D. Ross (2012). Gastrophryne carolinensis (Eastern Narrow-mouthed Toad). Predation. Herpetological Review 43(1):118. (PDF) (SREL Reprint #3212)
- Crawford, BA, CR Hickman, TM Luhring (2011). Testing the threat-sensitive hypothesis with predator familiarity and dietary specificity. Ethology 117:1-8. (PDF) (SREL Reprint #3193)
- Earl, JE, TM Luhring, BK Williams, RD Semlitsch (2011). Biomass export of salamanders and anurans from ponds is affected differentially by changes in canopy cover. Freshwater Biology 56:2473-2482. (PDF)
- Luhring, TM, JD Willson, CT Winne (2011). Nerodia fasciata. Inter-Wetland Movement. Herpetological Review 42:100-101. (PDF) (SREL Reprint #3181)
- Luhring, TM (2010). Siren sp (Siren) predation. Herpetological Review 41:470. (PDF)
- Schalk, CM, TM Luhring, BA Crawford (2010) Summer microhabitat use of the greater siren (Siren lacertina) and two-toed amphiuma (Amphiuma means) in an isolated wetland. Amphibia-Reptilia 31:251-256. (PDF) (SREL Reprint #3138)
- Luhring, TM, BD Todd (2010). Siren intermedia (Lesser Siren). Drought survival. Herpetological Review 41:60. (PDF) (SREL Reprint #3137)
- Schalk, CM, TM Luhring (2010). Vagility of aquatic salamanders: Implications for wetland connectivity. Journal of Herpetology 44:104-109. (PDF) (SREL Reprint #3132)
- Luhring, TM (2010). Predation by a Green Heron (Butorides virescens) on a Greater Siren (Siren lacertina). Bulletin of the Chicago Herpetological Society 45:33. (PDF) (SREL Reprint #3134)
- Schalk, CM, BA Crawford, TM Luhring (2009). Siren lacertina (greater siren) Predation. Bulletin of the Chicago Herpetological Society 44:56. (PDF)
- Luhring, TM (2009). Using PIT tags to evaluate non-individual-specific marks under field conditions: a case study with Greater Siren (Siren lacertina). Herpetological Review 40:170-173. (PDF)
- Todd, BD, TM Luhring, BB Rothermel, JW Gibbons (2009). Effects of forest removal on amphibian migrations: implications for habitat and landscape connectivity. Journal of Applied Ecology 46:554-561. (PDF) (SREL Reprint #3100)
- Gibbons, JW, TM Luhring (2009). Reptiles. Pages 497-505. In: G. E. Likens, ed. Encyclopedia of Inland Waters. Volume 3. Oxford: Elsevier. (PDF)
- Luhring, TM, CA Jennison (2008). A new stratified aquatic sampling technique for aquatic vertebrates. Journal of Freshwater Ecology 23:445-450. (PDF)(SREL Reprint #3347)
- Luhring, TM, GW Barrett (2008). Nesting ecology of the golden mouse: An oikos engineer. Pages 151-165. In: GW Barrett, GA Feldhamer, eds. The Golden Mouse: Ecology and Conservation. Springer. New York, NY.
- Todd, BD, BB Rothermel, RN Reed, TM Luhring, K Schlatter, T Trenkamp, JW Gibbons (2008). Habitat alteration increases invasive fire ant abundance to the detriment of amphibians and reptiles. Biological Invasions 10:539-546. (PDF) (SREL Reprint #3064)
- Luhring, TM (2008). “Problem species” of the Savannah River Site such as Brimley’s chorus frog, Pseudacris brimleyi, demonstrate the hidden biodiversity concept on an intensively-studied government reserve. Southeastern Naturalist 7:371-373. (PDF) (SREL Reprint #3067)
- Luhring, TM (2007). Reptiles and amphibians of boy scout camp Linwood-Hayne: Results from an undergraduate-initiated three year opportunistic inventory. Georgia Journal of Science 65:104-111. (PDF)(SREL Reprint #3057)
- Luhring, TM (2007). Siren lacertina (greater siren) Diet. Herpetological Review 38:317. (PDF) (SREL Reprint #3058)
- Luhring, TM, CA Young (2006). Innovative techniques for sampling stream salamanders. Herpetological Review 37:181-183. (PDF) (SREL Reprint #2967)
- Darley, M, A Howard, C Flaute, K Miller, TM Luhring, L Martin, C Ketter (2006). A laboratory manual for principals of biology II: Biology 1108L. Fourth Edition. Contemporary Publishing Company of Raleigh. Raleigh, NC.
- Rothermel, BB, TM Luhring (2005). Burrow availability and desiccation risk of mole salamanders (Ambystoma talpoideum) in harvested versus unharvested forest stands. Journal of Herpetology 39:619-626. (PDF) (SREL Reprint # 2913)